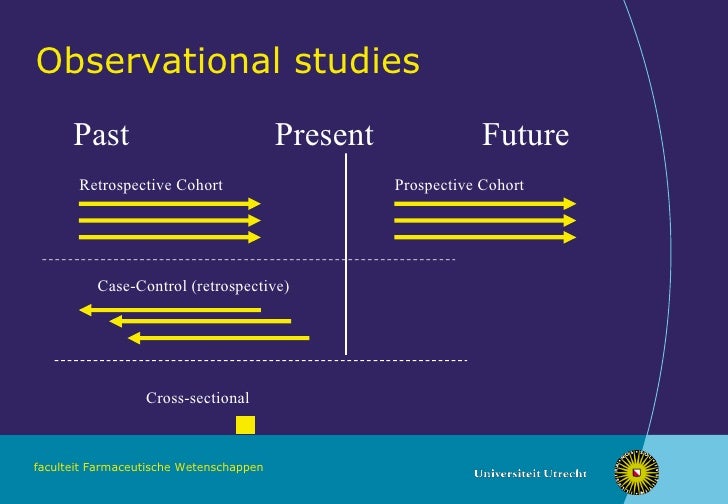  
The definition of the problem under study does not change once the data collection starts.Ī retrospective study is one in which you look backwards at data that have already been collected or generated, to answer a scientific (usually medical) problem. what you're measuring, who you're measuring, how many subjects, etc.), and then gathers data in the future in accordance with the design. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.A prospective study is one that identifies a scientific (usually medical) problem to be studied, specifies a study design protocol (e.g. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Supplemental material This content has been supplied by the author(s). Provenance and peer review Not commissioned externally peer reviewed. Patient and public involvement Patients and/or the public were not involved in the design, conduct, reporting or dissemination plans of this research. CL: grants from BMS, MSD, Novartis, Sanofi, Pierre Fabre consulting fees from BMS, MSD, Novartis, Amgen, Roche, Merck, Serono, Sanofi, Pierre Fabre and teaching fees from Roche, BMS, Novartis, Amgen, MSD. LM: grants from BMS, Novartis, Roche, MSD, GSK, Pierre Fabre and consulting fees from BMS, Novartis, Roche, MSD, GSK, Pierre Fabre. J-JG : consulting fees from MSD, Roche, Novartis, Amgen, Pierre-Fabre, Sanofi, Philogen, Merck, Pfizer. TL: consulting fees from MSD, BMS, Pierre Fabre, Novartis. FB-P: consulting fees from BMS and Sanofi. PS: consulting fees from Roche, Novartis, BMS, Pierre FABRE, MSD. Supervision: LP, SDalle, OD, ST, SDalac, DL, HM, J-PA, JDQ, PS, FB-P, TL, EM, FA, FG-B, J-JG, P-ES, CA, BO, CD, LM, CL.įunding This study did not receive external funding, but the MelBase cohort is sponsored by the French National Cancer Institute and by BMS, MSD, Novartis and Roche.Ĭompeting interests DL: fees from Novartis, BMS et MSD. Administrative, technical or material support: CA, CL. Critical revision of the manuscript for important intellectual content: LP, SDalle, OD, ST, SDalac, DL, HM, J-PA, JDQ, PS, FB-P, TL, EM, FA, FG-B, J-JG, P-ES, CA, BO, CD, LM, CL. Drafting of the manuscript: LP, CA, BO, CL. Acquisition, analysis or interpretation of data: LP, SDalle, OD, ST, SDalac, DL, HM, J-PA, JDQ, PS, FB-P, TL, EM, FA, FG-B, J-JG, P-ES, CA, BO, CD, LM, CL. LP accepts full responsibility for the work and/or the conduct of the study, had access to the data and controlled the decision to punblish. 21 Dermatology, Hopital Saint-Louis, Paris, Île-de-France, Franceĭr Léo Plaçais, Médecine Interne, Hospital Bicetre, Le Kremlin-Bicetre 94270, France Ĭontributors LP, CA, BO and CL had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.20 Dermatology, CHU Lille, Lille, Hauts-de-France, France.19 Dermatology, Centre Hospitalier Universitaire de Bordeaux, Bordeaux, Aquitaine, France.18 Dermatology, CHU Saint-Louis, Paris, Île-de-France, France.17 Dermatology, CHU Nimes, Nimes, Languedoc-Roussillon, France. 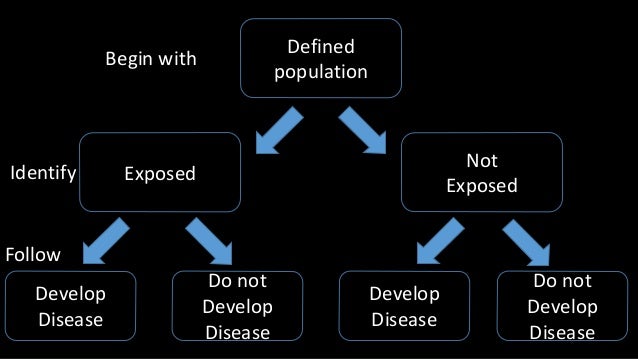
16 Dermatology, Hôpital de la Timone, Marseille, Provence-Alpes-Côte d'Azu, France.15 Dermatology, CHU de Nancy, Nancy, Lorraine, France.14 Dermatology, CHU Besancon, Besancon, France.13 Dermatology, Hopital Avicenne, Bobigny, France.12 Oncology, Centre Eugene Marquis, Rennes, France.11 Oncodermatology, Hopital Bichat - Claude-Bernard, Paris, France.10 Dermatology, Hôpital Ambroise Paré, Neuilly-sur-Seine, Île-de-France, France.9 Dermatology, Centre Hospitalier Annecy Genevois, Epagny Metz-Tessy, France.The goal of a retrospective audit is to do a ‘deep dive’ on the internal claims process. Retrospective audits involve reviewing claims that have already been submitted and preferably adjudicated as either paid, denied, or pending. 8 Dermatology, Centre Hospitalier Universitaire Amiens-Picardie, Amiens, Hauts-de-France, France The goal of a prospective audit is to catch any billing or coding errors before the claim is submitted.7 Dermatology, University Hospital, Nice, France.6 Dermatology, CHU Brest, Brest, Bretagne, France.5 Dermatology, CHU Dijon, Dijon, Bourgogne, France.4 Dermatology, CHU Grenoble Alpes, Grenoble, Auvergne-Rhone-Alpes, France.3 Dermatology, CHU Montpellier, Montpellier, Languedoc-Roussillon, France.2 Dermatology, Centre Hospitalier Universitaire de Lyon, Lyon, Rhône-Alpes, France.1 Internal Medicine and Clinical Immunology, Hopital Bicêtre, Le Kremlin-Bicêtre, Île-de-France, France.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
